Leiden Wall Formulas
Leiden has a rich history of physics discoveries. For instance, Kamerlingh Onnes, Lorentz and Zeeman earned the Nobel Prize for their discoveries of superconductivity and the Zeeman effect, while Snell's law of refraction, discovered in the 1600's, has been incorporated in study books all over the world. Physicists Sense Jan van der Molen and Ivo van Vulpen, inspired by the wonderful wall poems of the TEGENBEELD foundation Leiden's inner city, have strived for famous formulas to flaunt on the walls. They came to realize that you can sense the beauty of a formula even without fully understanding it, much like we feel the weight of words in foreign poetry, even if we can’t read them. The artists of TEGENBEELD have wholeheartedly joined the effort, by designing and painting original illustrations for each formula. In the end, the project will entail about ten wall formulas, including the seven that are already in place.
This project has inspired wall formula projects in other cities, including Utrecht. Go to www.utrechtsemuurformules.nl to see
the Utrecht wall formulas.
Einstein field equation Map
Albert Einstein (1879-1955)
Explanation: Einstein posed that every object (from an apple to a planet) curves the space around it. This means that objects attract each other--the famous law of gravity--but also bend light. Star light therefore travels past a heavy celestial body in a curved path. Einstein's formula--the so-called field equations of the theory of general relativity--describes how space gets deformed (left side) by an object within that space (right side). Wiki
Leiden component: Einstein was an extraordinary professor in Leiden and frequently resided here to collaborate with his Leiden colleagues. Although he didn't deduce this formula in Leiden, the third component--the cosmological constant 'Λ'--is a direct consequence of discussions with Leiden professor Willem de Sitter.
Snell's law Map
Willebrord Snel van Royen (Snellius) (1580-1626)
Explanation: A light ray changes direction in the transition to a different medium, for example from air to water. This creates the optical illusion of a snapped straw in a glass of lemonade. The breaking indices (n) of the respective media determine the angle (θ) of deflection. Wiki
Leiden component: Snell was a professor in Leiden. He did research in various scientific disciplines, including geometry and optics. He used triangulation to measure the Earth's circumference and he developed his famous law of refraction.
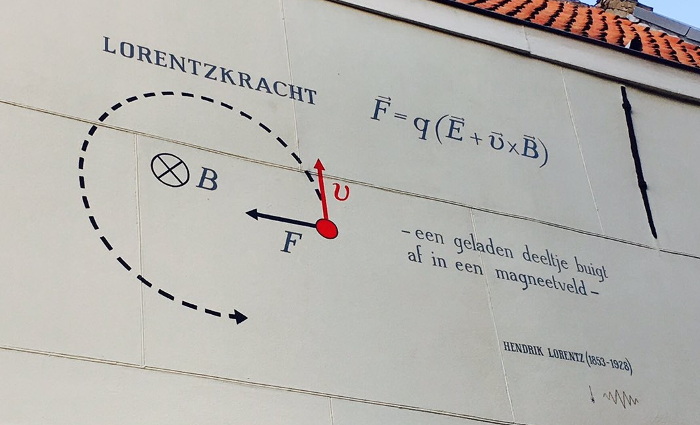
Lorentz force Map
Hendrik Lorentz (1853-1928)
Explanation: The Lorentz force (F) deflects a charged particle (q) within a magnetic field (B). Within an electric field (E), the particle additionally experiences an acceleration. Wiki
Leiden component: Lorentz was a physics professor in Leiden. He made a number of important discoveries here. He deduced for instance that objects become smaller as they reach the speed of light (Lorentz contraction) and described together with Pieter Zeeman the Zeeman effect, for which they received the Nobel Prize in 1902. He had already predicted this effect before with his Lorentz force.
Oort constants Map
Jan Oort (1900-1992)
Explanation: A minus B gives the Sun's angular velocity (vΘ/RΘ) around the center of the Milky Way. A plus B gives the decrease in speed (-[dv/dR]RΘ) of nearby stars as they are farther away from the center. This value turned out to be surprisingly low, and launched the search for the mysterious 'dark matter', which continues to this day. Scientists can determine the values of A and B from observations, contrary to their individual components. Wiki
Leiden component: Oort was an astronomy professor in Leiden. He discovered predicted the existence of the Oort cloud--a giant collection of small space objects revolving around the Sun in an orbit about 100,000 times as large as Earth's. Moreover, he formulated the Oort constants, from which he calculated the period of our Sun's orbit around the center of the Milky Way: more than 200 million years.
Lorentz contraction Map
Hendrik Lorentz (1853-1928)
Explanation: The actual length of an object is the original length at rest (L0) times a factor smaller than one, which is related to its speed (v). The faster an object travels, the smaller this so--called (inverse) Lorentz factor, and therefore the shorter its length. Or vice versa: the faster an observer is travelling, the smaller all stationary objects get. Einstein used the Lorentz contraction in his theory of special relativity. Wiki
Leiden component: Lorentz was physics professor in Leiden. Apart from the Lorentz contraction he made a number of other important discoveries. He described the Zeeman effect together with Pieter Zeeman, for which they received the Nobel Prize in 1902. Earlier, he discovered the Lorentz force, which provides a theoretical explanation for the Zeeman effect.
Electron spin Map
Samuel Goudsmit (1902-1978) -- George Uhlenbeck (1900-1988)
Explanation: Electrons are small particles orbiting an atomic nucleus. Apart from electrical charge and mass, they possess a third property which is crucial for our understanding of their behavior: spin. The only way to correctly describe spin is through quantum mechanics. However, you can imagine the concept by thinking of an electron that rotates--indeed spins--around its axis. The magnitude of an electron's spin is given by ℏ times a half. (ℏ is Planck's constant divided by 2𝜋.) The special phenomenon here is that electrons only come in two versions: they either rotate clockwise (spin down, or Sz=-1/2*ℏ), or counterclockwise (spin up, or Sz=+1/2*ℏ). Wiki
Leiden component: Samuel Goudsmit and George Uhlenbeck obtained their Master's and PhD degrees at Leiden University. Together they realized that electrons must have the property of spin, and that this can only take up two values (up and down).
Huygens' Pendulum Formula Map
Christiaan Huygens (1629-1695)
Explanation: Huygens' pendulum formula describes the connection between the length of a pendulum and the time that it takes to swing from left to right and back. This period, Huygens discovered, is only determined
by the length of the pendulum and gravity.
Wiki
Leiden component:
Christiaan Huygens studied law and mathematics at Leiden University from 1645 to 1647. In 1673, he published the formula that relates the period of the swing to the length of the pendulum. Accurate
timing had become very important for astronomy, navigation and also phyics itself. Huygens actually took a patent on his pendulum clock.
Van der Waals state equation Map
Johannes Diderik van der Waals (1837-1923)
Explanation: The Van der Waals equation describes the link between temperature (T), pressure (p) and volume (V) of a gas. R is the gas constant, and n is the number of gas molecules, expressed in moles. An equation that did this already existed, but it couldn't explain a number of measurements and phenomena, such as the condensation of fluid from a gas. The improved Van der Waals state equation could.
Van der Waals reasoned that the atoms or molecules in a gas occupy a certain volume. This is the term nb in the equation. Also, the particles can 'feel' each other: they attract or repel each other. These forces, later coined Van der Waals-forces, are accounted or by the term an2/V2. Wiki
Leiden component: Van der Waals described the equation in his PhD thesis 'Over de Continuïteit van den Gas- en Vloeistoftoestand' (On the Continuity of the Gas and Liquid State), which he defended in 1873 at Leiden University. In 1877, he was the first physics professor at the University of Amsterdam.
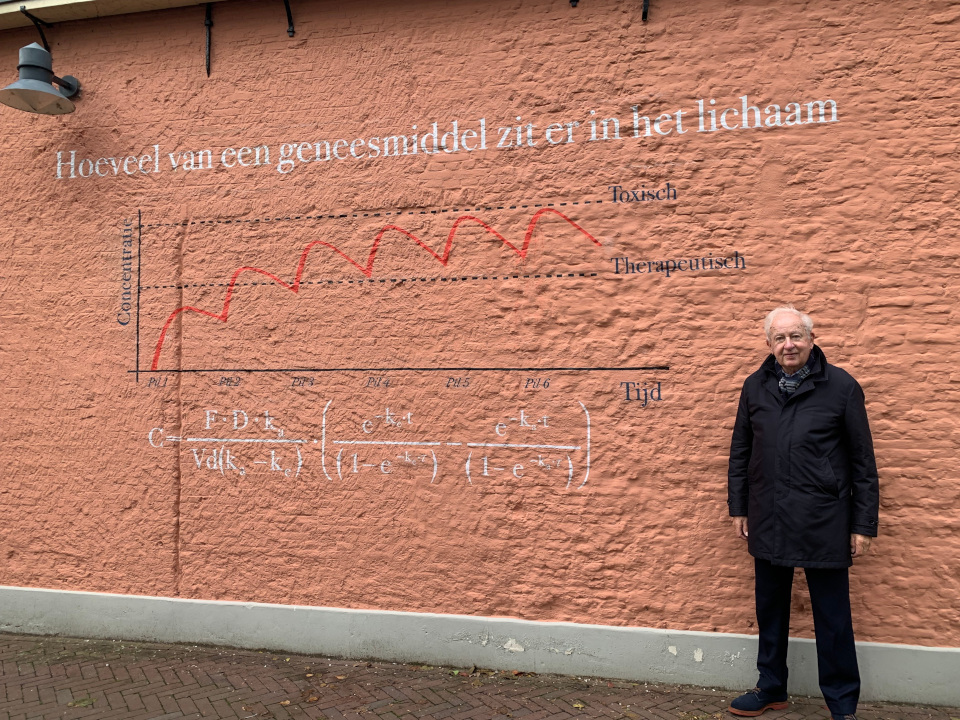
Pharmacokinetics Map
Douwe Breimer
Pharmacokinetics
Medicines work because the molecules in a tablet bind to certain proteins or enzymes in cells. The cells change their function and this is how medicines can lower the blood pressure or the cholesterol or can kill bacteria that cause an infection. For that to happen the molecules must be there in the right amount and at the right spot. The molecules in a pill are released when the tablet breaks apart in the stomach and are then absorbed in the blood. Liver and kidneys remove the molecules and all of this happens at a speed that is different for each medicine. Hence, the concentration of medicine will first go up and then go down. A pill taken next day will lead to a similar curve. This process can be described with this equation. When certain properties of the medicine are known and if you know how much must be in the blood for the medicine to work (therapeutic level) you can calculate the dose.
Leiden connection
To use equations like this one very precise measurements of the concentration of medicines in the blood must be possible. These techniques became available in the 1970?s and in Leiden Douwe Breimer and his group performed crucial research on the mathematical description of the time course of the concentration of medicines in blood and other body fluids, a field called pharmacokinetics. This research is still the basis of much of the work of the Leiden Academic Centre for Drug Research (LACDR) and the Centre for Human Drug Research (CHDR).
Professor Breimer was rector of Leiden University from 2001 to 2007.